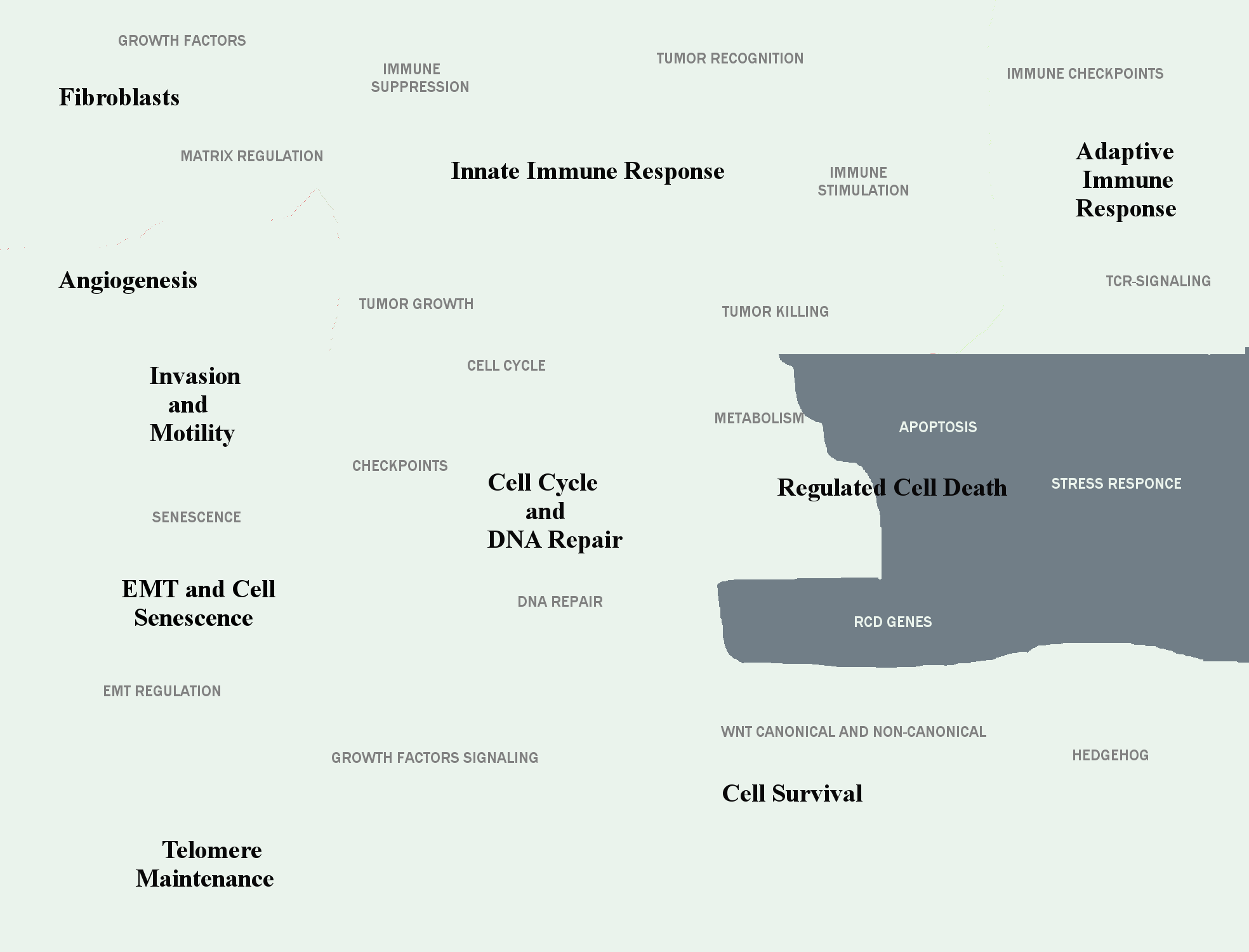
The hallmarks of cancer in ACSN 2.0
Sustaining Proliferative Signalling
Typically, cells of the body require hormones and other molecules that act as signals for them to grow and divide. Cancer cells, however, have the ability to grow without these external signals. There are multiple ways in which cancer cells can do this: by producing these signals themselves, known as autocrine signalling; by permanently activating the signalling pathways that respond to these signals; or by destroying 'off switches' that prevents excessive growth from these signals (negative feedback). In addition, cell division in normal, non-cancerous cells is tightly controlled. In cancer cells, these processes are deregulated because the proteins that control them are altered, leading to increased growth and cell division within the tumor
In ACSN:
Cell Survival map
- Growth Factors
- Hedgehog
- MAPK
- PI3K-AKT-mTOR
- WNT Canonical
- WNT Non-Canonical

Evading Growth Suppressors
The cell cycle clock. Cells do not divide in G0 and are quiescent. After receiving growth factor signals, they prepare for division by entering G1, where everything within the cell except DNA is doubled. This doubling includes the size of the cell. The next phase of the cell cycle is S (synthesis) phase. It is the cell cycle phase where the chromosomes (DNA) are duplicated in preparation for cellular division. The transition from G1 to S is a checkpoint. If the cell has damaged DNA or is expressing oncogenes or other inappropriate proteins, specialized checkpoint proteins, tumor suppressors such as p53 or pRB, will interrupt the transition to S phase until the damage is repaired. If the damage cannot be repaired, the cell will initiate apoptosis, often referred to as cellular suicide, which is programmed cell death. If the tumor suppressor genes incur loss-of-function mutations or are knocked out, the damaged cell can continue to divide unchecked — one of the hallmarks of cancer.
In ACSN:
Cell Cycle and DNA repair map
- G1-CC phase
- G1-S checkpoint
- G2-CC phase
- G2-M checkpoint
- M-CC phase
- S-CC phase
- S phase checkpoint
- Spindle checkpoint

Resisting Cell Death
Cells have the ability to 'self-destruct'; a process known as apoptosis. This is required for organisms to grow and develop properly, for maintaining tissues of the body, and is also initiated when a cell is damaged or infected. Cancer cells, however, lose this ability; even though cells may become grossly abnormal, they do not apoptose. The cancer cells may do this by altering the mechanisms that detect the damage or abnormalities. This means that proper signalling cannot occur, thus apoptosis cannot activate. They may also have defects in the downstream signalling itself, or the proteins involved in apoptosis, each of which will also prevent proper apoptosis.
In ACSN:
Regulated Cell Death map
- Antioxidant Response
- Apoptosis
- Caspases
- Death Receptor Pathways
- Dependence Receptors
- ER Stress
- FAS Response
- Ferroptosis
See more|Hide
Enabling Replicative Immortality
Cells of the body don't normally have the ability to divide indefinitely. They have a limited number of divisions before the cells become unable to divide (senescence), or die (crisis). The cause of these barriers is primarily due to the DNA at the end of chromosomes, known as telomeres. Telomeric DNA shortens with every cell division, until it becomes so short it activates senescence, so the cell stops dividing. Cancer cells bypass this barrier by manipulating enzymes that increase the length of telomeres. Thus, they can divide indefinitely, without initiating senescence.
In ACSN:
Telomere Maintenance map
- Replication
- Shelterin
- Telomerase

Inducing Angiogenesis
Normal tissues of the body have blood vessels running through them that deliver oxygen from the lungs. Cells must be close to the blood vessels to get enough oxygen for them to survive. New blood vessels are formed during the development of embryos, during wound repair and during the female reproductive cycle. An expanding tumour requires new blood vessels to deliver adequate oxygen to the cancer cells, and thus exploits these normal physiological processes for its benefit. To do this, the cancer cells acquire the ability to orchestrate production of new vasculature by activating the 'angiogenic switch'. In doing so, they control non-cancerous cells that are present in the tumor that can form blood vessels by reducing the production of factors that inhibit blood vessel production, and increasing the production of factors that promote blood vessel formation.
In ACSN:
Agiogenesis map

Activating Invasion and Metastasis
One of the most well known properties of cancer cells is their ability to invade neighboring tissues. It is what dictates whether the tumor is benign or malignant, and is the reason for their dissemination around the body. The cancer cells have to undergo a multitude of changes in order for them to acquire the ability to metastasize. It is a multistep process that starts with local invasion of the cells into the surrounding tissues. They then have to invade blood vessels, survive in the harsh environment of the circulatory system, exit this system and then start dividing in the new tissue.
In ACSN:
EMT and Senescence map
- Adherens junctions
- Cell-Cell Adhesions
- Cell Matrix Adhesions
- Cytoskeleton polarity
- Desmosomes
- ECM
- EMT regulators
- Lysosome-Endosome
- Mitochondria oxidative stress
- Senescence
- Tight junctions
Invasion and motility map
Cancer-Associated Fibroblast map
- CAF inhibition antitumor
See more|Hide

Genome Instability and Mutation
Cancer cells generally have severe chromosomal abnormalities which worsen as the disease progresses. HeLa cells, for example, are extremely prolific and have tetraploidy 12, trisomy 6, 8, and 17, and a modal chromosome number of 82 (rather than the normal diploid number of 46).[22] Small genetic mutations are most likely what begin tumorigenesis, but once cells begin the breakage-fusion-bridge (BFB) cycle, they are able to mutate at much faster rates.
In ACSN:
Cell Cycle and DNA repair map
- A-NHEJ
- BER
- C-NHEJ
- DR
- Fanconi
- HR
- MMR
- NER
- SSA
- TLS

Tumor-Promoting Inflammation
Recent discoveries have highlighted the role of local chronic inflammation in inducing many types of cancer. Inflammation leads to angiogenesis and more of an immune response. The degradation of extracellular matrix necessary to form new blood vessels increases the odds of metastasis.
In ACSN:
CAF cell map
- CAF inhibition antitumor
- CORE
- Cytokines-Chemokines production
- Fibroblast Activation Markers
- Growth Factors production
- Growth Factors signaling pathways
See more|Hide

Reprogramming Energy Metabolism
Most cancer cells use abnormal metabolic pathways to generate energy, a fact appreciated since the early twentieth century with the postulation of the Warburg hypothesis, but only now gaining renewed research interest. Cancer cells exhibiting the Warburg effect upregulate glycolysis and lactic acid fermentation in the cytosol and prevent mitochondria from completing normal aerobic respiration (oxidation of pyruvate, the citric acid cycle, and the electron transport chain). Instead of completely oxidizing glucose to produce as much ATP as possible, cancer cells would rather convert pyruvate into the building blocks for more cells. In fact, the low ATP:ADP ratio caused by this effect likely contributes to the deactivation of mitochondria. Mitochondrial membrane potential is hyperpolarized to prevent voltage-sensitive permeability transition pores (PTP) from triggering of apoptosis.
In ACSN:
Regulated Cell Death map
- Glucose Metabolism
- Glutamine Metabolism
- Mitochondrial Genes
- Mitochondrial Metabolism
- Mitochondrial Permeability Transition
- Oxidative Phosphorylation and TCA cycle
- Pentose Phosphate Pathway
- Porphyrin Metabolism
RECON

Evading Immune Destruction
Despite cancer cells causing increased inflammation and angiogenesis, they also appear to be able to avoid interaction with the body’s via a loss of interleukin-33 immune system.
In ACSN:
Adaptive Immuninty map
- Activating checkpoints
- Inhibiting checkpoints
- SMAC
- TRC signaling
Innate Immunity map
- Antigen presentation and Immunostimulatory checkpoints
- Danger Signal pathways
- FC Receptors
- Immunostimulatory core pathways
- Immunostimulatory Cytokine expression
- Immunostimulatory Cytokine pathways
See more|Hide